Atomic StructureHard
Question
If the total energy of an electron in hydrogen like atom in an excited state is -3.4 eV, then the
de-Broglie wavelength of the electron is
de-Broglie wavelength of the electron is
Options
A.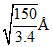
B.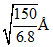
C.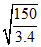 nm
nm
D.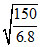 nm
nm
More Atomic Structure Questions
Suppose that means were available for stripping 29 electrons from 30Zn in vapours of this metal. The ionization energy f...A photon of 2.55 eV is emitted out by an electronic transition in hydrogen atom. The change in de Broglie wavelength of ...Which one is a wrong statement ?...When β-particles are sent through a tin metal foil, most of them go straight through the foil as...A certain molecule has an energy level diagram for its vibrational energy in which two levels are 0.0141 eV apart. The w...