NEET | 2018Atomic StructureHard
Question
Which one is a wrong statement ?
Options
A.
Total orbital angular momentum of electron in 's' orbital is equal to zero
B.
An orbital is designated by three quantum numbers while an electron in an atom is designated by four quantum numbers.
C.
The electronic configuration of N atom is
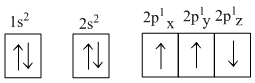
D.
The value of m for is zero
Solution
The correct configuration of 'N' is

Create a free account to view solution
View Solution FreeMore Atomic Structure Questions
The pair of electron in the given carbanion, CH3C ≡C⊝is present in which of the following orbitals?...Small packets of light is called...Which of the following is electronic configuration of Cu2+ (Z = 29) ?...The dynamic mass (in kg) of the photon with a wavelength corresponding to the series limit of the Balmer transitions of ...The e/m ratio of a particle of charge 2 unit and mass 4 amu is...