Ionic EquilibriumHard
Question
At 25oC, the solubility product of Mg(OH)2 is 1.0 × 10-11. At which pH, will Mg2+ ions start precipitating in the form of Mg(OH)2 from a solution of 0.001 M Mg2+ ions ?
Options
A.9
B.10
C.11
D.8
Solution
Mg2+ + 2OH- ⇋ Mg(OH)2
Ksp = [Mg2+][OH-]2
[OH-]=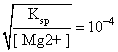
∴ pOH = 4 and pH = 10
Ksp = [Mg2+][OH-]2
[OH-]=
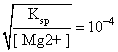
∴ pOH = 4 and pH = 10
Create a free account to view solution
View Solution FreeMore Ionic Equilibrium Questions
0.1 millimole of CdSO4 are present in 10 mL acid solution of 0.08 N HCl. Now H2S is passed to precipitate all the Cd2+ i...Which is the correct statement for the given acids?...Which one of the following substances has the highest proton affinity?...Aniline behaves as a weak base. When 0.1 M, 50 ml solution of aniline was mixed with 0.1 M, 25 ml solution of HCl the pH...The sodium salt of a certain weak monobasic organic acid is hydrolysed to an extent of 3% in its 0.1M solution at 25oC. ...