Ionic EquilibriumHard
Question
The sodium salt of a certain weak monobasic organic acid is hydrolysed to an extent of 3% in its 0.1M solution at 25oC. Given that the ionic product of water is 10-14 at this temperature, what is the dissociation constant of the acid?
Options
A.≈ 1 × 10-10
B.≈ 1 × 10-9
C.3.33 × 10-9
D.3.33 × 10-10
Solution
h = .03 C = 0.1 M
Kh = Ch2 = 9 × 10-5
Kh =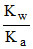 = 9 × 10-5 ⇒ Ka =
= 9 × 10-5 ⇒ Ka = 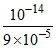 = 1.11 × 10-10 ≈ 1 × 10-10
= 1.11 × 10-10 ≈ 1 × 10-10
Kh = Ch2 = 9 × 10-5
Kh =
Create a free account to view solution
View Solution FreeMore Ionic Equilibrium Questions
A certain sample of rainwater gives a yellow colour with methyl red [pH range 4.2 (red) – 6.2 (yellow)] and a yellow col...1 M NaCl and 1 M HCl are present in an aqueous solution. The solution is...Arsenic (III) sulphide form a sol with negative charge. Which of the following ionic substances will have lowest floceul...A 2.5 g impure sample containing weak monoacidic base (Molecular weight = 45) is dissolved in 100 ml water and titrated ...The compound that is not a Lewis acid is :...