ElectrochemistryHard
Question
Consider the cell 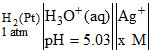 Ag. The measured EMF of the cell is 1.0 V. What is the value of x ? EoAg+,Ag = + 0.8 V. [T = 25oC]
Ag. The measured EMF of the cell is 1.0 V. What is the value of x ? EoAg+,Ag = + 0.8 V. [T = 25oC]
Options
A.2 × 10-2 M
B.2 × 10-3 M
C.1.5 x 10-3 M
D.1.5 × 10-2 M
Solution
H2(Pt) (1 atm) || | Ag+(xM) | Ag
1.0 = (0 + 0.8) -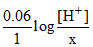
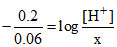
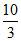 = pH + log x
= pH + log x
log x = - 1.7
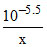 = 1.62 × 10-4
= 1.62 × 10-4
x = 2 × 10-2 M
1.0 = (0 + 0.8) -
log x = - 1.7
x = 2 × 10-2 M
Create a free account to view solution
View Solution FreeMore Electrochemistry Questions
Which cannot displace hydrogen from its compound ?...Time required to deposit one millimole of aluminum metal by the passage of 9.65 amperes through aqueous solution of alum...Given : Hg22+ + 2e → 2Hg , Eo = 0.789 V & Hg2+ + 2e → Hg, Eo = 0.854 V, calculate the equilibrium constant f...Cu+ + e- → Cu, Eo = x1 volt ;Cu2+ + 2e- Cu, Eo = x2 volt, then forCu2+ + e- Cu+, Eo (volt) will be -...Ostwald′s dilution law is application on :...