ElectrochemistryHardBloom L3
Question
The cell Zn|Zn²⁺(1M)||Cu²⁺(1M)|Cu with $E^\circ_{\text{cell}} = 1.10$ V was allowed to be completely discharged at 298 K. The relative concentration of Zn²⁺ to Cu²⁺, $\frac{[\text{Zn}^{2+}]}{[\text{Cu}^{2+}]}$, is 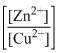
Options
A.antilog(24.08)
B.37.3
C.$10^{37.3}$
D.$9.65 \times 10^4$
Solution
{"given":"A galvanic cell with Zn|Zn²⁺(1M)||Cu²⁺(1M)|Cu configuration, standard cell potential $E^\\circ_{\\text{cell}} = 1.10$ V, temperature T = 298 K, and the cell is completely discharged meaning final cell potential $E_{\\text{cell}} = 0$ V. The cell reaction is Zn + Cu²⁺ → Zn²⁺ + Cu with n = 2 electrons transferred.","key_observation":"At complete discharge, the cell reaches equilibrium where $E_{\\text{cell}} = 0$. Using the Nernst equation: $E_{\\text{cell}} = E^\\circ_{\\text{cell}} - \\frac{0.059}{n} \\log Q$, where Q is the reaction quotient $\\frac{[\\text{Zn}^{2+}]}{[\\text{Cu}^{2+}]}$ and n = 2 electrons transferred in the overall cell reaction. At equilibrium, we can solve for the concentration ratio using logarithmic relationships.","option_analysis":[{"label":"(A)","text":"antilog(24.08)","verdict":"incorrect","explanation":"This would give $10^{24.08}$ which is not the correct value. The calculation gives log Q = 37.3, so the answer should be $10^{37.3}$, not $10^{24.08}$."},{"label":"(B)","text":"37.3","verdict":"incorrect","explanation":"This is the logarithmic value, not the actual concentration ratio. We calculated log([Zn²⁺]/[Cu²⁺]) = 37.3, but the question asks for the ratio itself, which is $10^{37.3}$."},{"label":"(C)","text":"$10^{37.3}$","verdict":"correct","explanation":"Step 1: At complete discharge, $E_{\\text{cell}} = 0$:\n$$0 = E^\\circ_{\\text{cell}} - \\frac{0.059}{n} \\log Q$$\nStep 2: Substituting values:\n$$0 = 1.10 - \\frac{0.059}{2} \\log \\frac{[\\text{Zn}^{2+}]}{[\\text{Cu}^{2+}]}$$\nStep 3: Solving for the logarithm:\n$$\\log \\frac{[\\text{Zn}^{2+}]}{[\\text{Cu}^{2+}]} = \\frac{1.10 \\times 2}{0.059} = 37.3$$\nStep 4: Taking antilog:\n$$\\frac{[\\text{Zn}^{2+}]}{[\\text{Cu}^{2+}]} = 10^{37.3}$$"},{"label":"(D)","text":"$9.65 \\times 10^4$","verdict":"incorrect","explanation":"This is a much smaller value than the correct answer. The calculation clearly shows that the ratio should be $10^{37.3}$, which is an enormously large number, not $9.65 \\times 10^4$."}],"answer":"(C)","formula_steps":[]}
Create a free account to view solution
View Solution FreeMore Electrochemistry Questions
The standard potentials of MnO4−|Mn2+ and MnO2|Mn2+ electrodes in acid solution are 1.51 and 1.23 V, respectively. Stand...Select the correct option if it is known that Ksp (AgCl) > Ksp (AgBr) > Ksp (AgI)...A dilute aqueous solution of Na2SO4 is electrolysed using platinum electrodes. The products at the anode and cathode are...When iron is rusted, it is :...Both oxidation and reduction takes place in :...