ElectrochemistryHard
Question
A current of 2.0 ampere passed for 5 hours through a molten tin salt deposits 22.2 gm of metal. The oxidation number of metal would be : (Sn = 118) :-
Options
A.+ 2
B.+ 4
C.+ 3
D.+ 1
Solution
W = 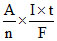
A = atomic mass
n = valancy of ion (Sn+n)
t = time of current flow in seconds.
A = atomic mass
n = valancy of ion (Sn+n)
t = time of current flow in seconds.
Create a free account to view solution
View Solution FreeMore Electrochemistry Questions
A line L : y = mx + 3 meets y - axis at E(0,3) and the arc of the parabola y2 = 16x, 0 ≤ y ≤ 6 at the point ...Three moles of electrons are passed through three solutions in succession containing AgNO3, CuSO4, and AuCl3, respective...Given EoAg+/Ag = 0.80V, EoMg2+/Mg = - 2.37V, EoCu2+/Cu = 0.34, EoHg2+/Hg = 0.79 V.Which of the following statements is/a...A dilute aqueous solution of Na2SO4 is electrolysed using platinum electrodes. The products at the anode and cathode are...The standard reduction potentials at 298 K for the following half reactions are given against each Zn2+(aq) + 2e ⇋...