NEETThermodynamicsHard
Question
6 mole of an ideal gas expand isothermal and reversibly from a volume of 1 liter to avolume of 10 liters at 27oC. What is the maximum work done ?
Options
A.47 J
B.100 kJ
C.0
D.34.365kJ
Solution
W = - 2 2.303 nRT log 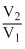
GIven n = 6, T = 27oC = 273 = 27 = 300 K V1 = 1L, V2 = 10L
∴ W = - 22.303 × 6 × 8.314 × 300log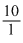
= 34.465kJ
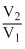
GIven n = 6, T = 27oC = 273 = 27 = 300 K V1 = 1L, V2 = 10L
∴ W = - 22.303 × 6 × 8.314 × 300log
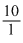
= 34.465kJ
Create a free account to view solution
View Solution FreeMore Thermodynamics Questions
For the reaction of the one mole of zinc dust with one mole of sulphuric acid in a bombcalorimeter, ᐃU and w corre...For the cyclic process given below, which of the following relations are correct?...What is the free energy change, ΔG, when 1.0 mole of water at 100°C and 1 atm pressure is converted into steam at 100°C ...The value of log10K for a reaction A ⇋ B is ( Given : áƒfHo298K = - 54.07 kJ mol-1, áƒrSo298K = 10JK-1mol-1 and R = ...Which of the following statement(s) is/are correct regarding ideal gas?...