SolutionHard
Question
The degree of dissociation (α) of a weak electrolyte, Ax By is related to van′t Hoff factor (i) by the expression :
Options
A.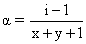
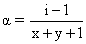
B.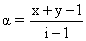
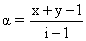
C.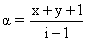
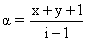
D.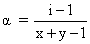
Solution
i = 1 - α + n α = 1 + α(n - 1)
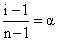
Ax By → xA+y + yB-x
n = x + y
So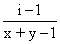
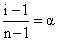
Ax By → xA+y + yB-x
n = x + y
So
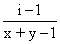
Create a free account to view solution
View Solution FreeMore Solution Questions
The vapour pressure of an aqueous solution is found to be 750 torr at a temperature T, and the same solution shows an el...Among the following mixtures, dipole-dipole as the major interaction, is present in...Following solutions were prepared by mixing different volumes of NaOH and HCl of different concentrations : a .60mLM10HC...Ratio of ᐃTb / Kb of 6% AB2 and 9% A2B (AB2 and A2B both are non-electrolytes) is 1 mol/kg in both cases. Hence, a...Based on the given diagram, which of the following statements regarding the solutions of two miscible volatile liquids a...