SolutionHard
Question
Based on the given diagram, which of the following statements regarding the solutions of two miscible volatile liquids are correct ?

Options
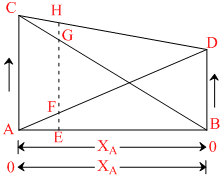
A.Plots AD and BC show the Raoult′s law is obeyed for the solution in which B is a solvent and A is the solute and as well as for that in which A is solvent and B is solute
B.Plots CD shows that Dalton′s law of partial pressures is obeyed by the binary solutions of components A and B
C.EF + EG = EH and AC and BD correspond to the vapour pressure of the pure solvent A and B respectively
D.As B is added to the component A, vapour pressure of A as well as of B changes
Solution
(A), (C) and (D) all are facts except (B). Because plot CD shows the total vapour pressure.
For ideal solution,
AD and AC obey Raoult′s law.
CD show Dalton′s law of partial pressure.
AC : PA0 XB = 0 ; XA = 1.0
BD : PB0 XB = 0 ; XB = 1.0
For ideal solution,
AD and AC obey Raoult′s law.
CD show Dalton′s law of partial pressure.
AC : PA0 XB = 0 ; XA = 1.0
BD : PB0 XB = 0 ; XB = 1.0
Create a free account to view solution
View Solution FreeMore Solution Questions
The vapour pressures of pure liquids A, B and C are 75, 22 and 10 torr, respectively. Which of the following is/are poss...For a dilute solution having molality m of a given solute in a solvent of mol.wt. M, b.pt. Tb and heat of vaporisation p...When the depression in freezing point is carried out, the equilibrium exist between...Temperature does not affect :...During depression of freezing point in a solution the following are in equilibrium :...