SolutionHard
Question
Ratio of ᐃTb / Kb of 6% AB2 and 9% A2B (AB2 and A2B both are non-electrolytes) is 1 mol/kg in both cases. Hence, atomic masses of A and B are respectively :
Options
A. 60, 90
B.40, 40
C.40, 10
D.10, 40
Solution
ᐃT = 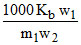
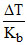 (AB2) =
(AB2) =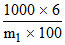 = 1
= 1
∴ m1 (AB2) = 60 = A + 2B
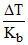 (A2B) =
(A2B) = 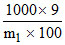 = 1
= 1
∴ m1 (A2B) = 90 = 2A + B
∴ A = 40, B = 10
∴ m1 (AB2) = 60 = A + 2B
∴ m1 (A2B) = 90 = 2A + B
∴ A = 40, B = 10
Create a free account to view solution
View Solution FreeMore Solution Questions
The normal freezing point of nitrobenzene is 278.82 K. A non-volatile solute is dissolved in it and a solution of molali...Which has maximum osmotic pressure at temperature T :...The freezing point depression of 0.001 m Kx[Fe(CN)6] is 7.10 × 10-3 K. Determine the value of x ? (Given Kƒ = ...The vapour pressure of a dilute aqueous solution of glucose is 750 mm of mercury at 373 K. The mole fraction of solute i...If sodium sulphate is considered to be completely dissociated into cations and anions in aqueous solution, the change in...