KTGHard
Question
Three perfect gases at absolute temperatures T1, T2 and T3 are mixed. The masses of molecules are m1, m2 and m3 and the number of molecules are n1, n2 and n3 respectively. Assuming no loss of energy, the final temperature of the mixture is :
Options
A.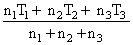
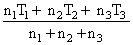
B.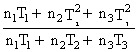
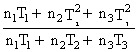
C.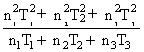
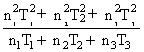
D.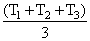
Solution
Data ⇒ n1T1k1 + n2kT2 + n2kT3 = (n1 + n2 + n3) kT
∴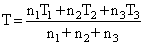
∴
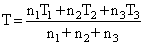
Create a free account to view solution
View Solution FreeMore KTG Questions
The internal energy of an ideal gas depends on -...N(< 100) molecules of a gas have velocities 1, 2, 3........ N km/s respectively. Then...In a sample of an ideal gas the average momentum of a molecule depends on...Statement A : Cp - Cv = R ; B: = = 1.67...A thermally insulated chamber of volume 2V0 is divided by a frictionless piston of area S & mass m into two equal parts ...