KTGHard
Question
N(< 100) molecules of a gas have velocities 1, 2, 3........ N km/s respectively. Then
Options
A.rms speed and average speed of molecules is same.
B.ratio of rms speed to average speed is √(2N + 1)(N + 1)/6N
C.ratio of rms speed to average speed is √(2N + 1)(N + 1)/6
D.ratio of rms speed to average speed of molecules is 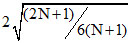
Solution
vrms = 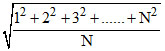 ⇒ vrms =
⇒ vrms = 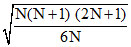
vrms =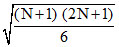 ...........(i)
...........(i)
var =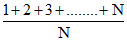
var =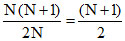 .........(ii)
.........(ii)
Form (i) & (ii)
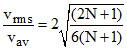
vrms =
var =
var =
Form (i) & (ii)
Create a free account to view solution
View Solution FreeMore KTG Questions
At constant pressure hydrogen is having temperature of 327o C . Till what temperature it is to be cooled so that the rms...Mole of H2 at 47oC and at pressure 4 atmosphere is mixed with 2 mole of N2 at 127o and at 5 atmosphere such that the vol...The pressure of an ideal gas is written as E = . Here E stands for...If the ratio of specific heats is γ and R is the gas constant then what will be the value of specific heat for gas ...The ratio ofat a given temperature is :-...