KTGHard
Question
Statement A : Cp - Cv = R ; B: = 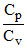 = 1.67
= 1.67
Options
A.A and B statement are true for all ideal gases.
B.A is true for all ideal gases and B is true for only monoatomic gases
C.A and B are true for monoatomic gases.
D.A is true for all ideal gases and B is true for only diatomic gases
More KTG Questions
In vanderwall′s equation, (V − B) = RT, the dimension of a is :-...Average kinetic energy of a gas molecule depends on-...Boiling water is changing into steam. Under this condition, the specific heat of water is...In given figure, let W1 and W2 be the workdone by the system in process A and B respectively then (initial and final vol...For an ideal gas molecules the following quantities having zero average values are....