Atomic StructureHard
Question
For an electron in the second orbit of hydrogen, the moment of momentum as per Bohr′s model is
Options
A.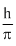
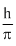
B.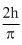
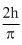
C.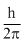
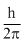
D.2πh
Solution
The moment of momentum is also known as angular momentum is also known Weangular momentum of electron.
angular momentum = n.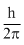
For second orbit n = 2
So, angular momentum = 2 ×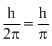 is the answer.
is the answer.
angular momentum = n.
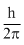
For second orbit n = 2
So, angular momentum = 2 ×
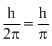 is the answer.
is the answer.Create a free account to view solution
View Solution FreeMore Atomic Structure Questions
The shortest wavelength of X-rays emitted from an X-ray tube depends on...If a graph of Binding energy (B/A) per nucleon versus mass number (A) looks like as shown in figure. Then using the curv...The transtion from the state n = 4 to n = 3 in a hydrogen like atom results in ultraviolet radiation. Infrared radiation...A radiation of energy E falls normally on a perfectly reflecting surface. The momentum transferred to the surface is...The transition from the state n = 3 to n = 1 in a hydrogen like atom results in ultraviolet radiation.Infrared radiation...