ThermodynamicsHard
Question
A mass of diatomic gas (γ ) at a pressure of 2 atmospheres is compressed adiabati cally so that its temperature rises from 27oC to 927oC . The pressure of the gas in the final state is
Options
A.8 atm
B.28 atm
C.68.7 atm
D.256 atm
Solution
For an adiabatic process
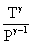 = constant
= constant
∴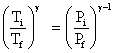
Pf = Pi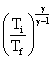 .......(i)
.......(i)
Here, Ti = 27oC = 300 K
Tf = 927oC = 1200 K
Pi = 2 atm
γ = 1.4
Substituting these values in eqn (i), we get
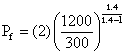
Pf = (2)(4)1.4/0.4 = 2(2)7/2 = (2)(2)7 = 28 = 256 atm
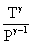 = constant
= constant∴
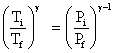
Pf = Pi
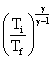 .......(i)
.......(i)Here, Ti = 27oC = 300 K
Tf = 927oC = 1200 K
Pi = 2 atm
γ = 1.4
Substituting these values in eqn (i), we get
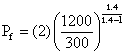
Pf = (2)(4)1.4/0.4 = 2(2)7/2 = (2)(2)7 = 28 = 256 atm
Create a free account to view solution
View Solution FreeMore Thermodynamics Questions
The density (ρ) of and ideal gas varies with temperature T as shown in figure. Then...A monoatomic ideal gas, initially at temperature T1, is enclosed in a cylinder fitted with a frictionless piston. The ga...When 1 kg of ice at 0oC melts to water at 0oC, the resulting change in its entropy, taking latent heat of ice to be 80 c...The amount of heat energy required to raise the temperature of 1 g of Helium at NTP, from T1 K to T2 K is :-...One mole of an ideal gas in initial state A undergoes a cyclic processABCA, as shown in the figure. Its pressure at A is...