NEET | 2013ThermodynamicsHard
Question
The amount of heat energy required to raise the temperature of 1 g of Helium at NTP, from T1 K to T2 K is :-
Options
A.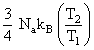
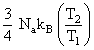
B.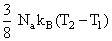
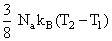
C.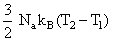
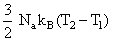
D.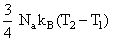
Solution
Number of moles in 1g He = 1/4
Amount of heat energy required to raise its temepratre from T1 K to T2 K
= nCv ᐃT
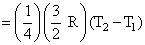
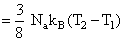
Amount of heat energy required to raise its temepratre from T1 K to T2 K
= nCv ᐃT
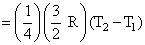
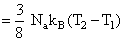
Create a free account to view solution
View Solution FreeMore Thermodynamics Questions
A carnot′s engine works between temperature 772oC and 27oC, the efficienty of the engine is :...One mole of an ideal atomic gas is heated at a constant pressure of one atmosphere from 0oC to 100oC. Then the change in...In a process, 500 calories of heat is given to a system and at the same time, 100 joules of work is done on the system. ...The curves shown represent adiabatic curves for monoatomic, diatomic & polyatomic (γ = 4/3) gases. The slopes for c...One mole of diatomic ideal gas undergoes a cyclic process ABC as shown in figure. The process BC is adiabatic. The tempe...