ThermodynamicsHard
Question
When 1 kg of ice at 0oC melts to water at 0oC, the resulting change in its entropy, taking latent heat of ice to be 80 cal/ oC, is
Options
A.273 cal /K
B.8 × 104 cal /K
C.80 cal /K
D.293 cal / K
Solution
Heat required to melt 1 kg ice at 0oC to water at 0oC is
Q = miceLice = (1 kg) (80 cal/g)
= (1000 g) (80 cal/g) = 8 × 104 cal
Change in entrophy, ᐃS =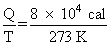 = 293 cal/K
= 293 cal/K
Note : In the equstion paper unit of latent heat of ice is given to be cal/ oC. It is wrong. The unit of latent heat of ice is cal/g.
Q = miceLice = (1 kg) (80 cal/g)
= (1000 g) (80 cal/g) = 8 × 104 cal
Change in entrophy, ᐃS =
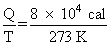 = 293 cal/K
= 293 cal/KNote : In the equstion paper unit of latent heat of ice is given to be cal/ oC. It is wrong. The unit of latent heat of ice is cal/g.
Create a free account to view solution
View Solution FreeMore Thermodynamics Questions
The ratio of the speed of sound in nitrogen gas to that in helium gas, at 300 K is...A refrigerator with coefficient of performance 1/3 releases 200 J of heat to a hot reservoir, then the work done on the ...In a process on a closed system of ideal gas, the initial pressure and volume is equal to the final pressure and volume...During the melting of a slab of ice at 273 K at atomospheric pressure...During the adiabatic expansion of two moles of a gas the internal energy of a gas found to decreases by 2 joule. The wor...