Heat and Thermal ExpansionHard
Question
A vessel is partitioned two equal halves by a fixed diathermy separator. Two different ideal gasses are filled in left (L) and right halves. The rms speed of the molecules in l part is equal to the mean speed of molecules in the R part. Then the ratio of a molecule in L part to that of a molecules in R part is : -
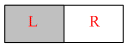

Options
A.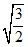
B.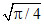
C.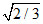
D.2π/8
More Heat and Thermal Expansion Questions
The density in grams per litre of ethylene (C2H4) at STP is : -...n moles of an ideal triatomic linear gas undergoes a proses in which the temperature changes with volume as T = k1V2 whe...A body cools in a surrounding which at a constant temperature of θ0 Assume that it obeys Newton′s law of cool...Three rods of the same dimensions have thermal conductivities 3k, 2k and k. They are arranged as shown, with their ends ...The reversible expansion of an ideal gas under adiabatic and isothermal conditions is shown in the figure. Which of the ...