Heat and Thermal ExpansionHard
Question
n moles of an ideal triatomic linear gas undergoes a proses in which the temperature changes with volume as T = k1V2 where k1 is a constant. Choose incorrect alternative :-
Options
A.At normal temperature Cv = 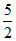 R
R
B.At any temperature Cp - Cv = R
C.At normal temperature molar heat capacity C = 3R
D.At any temperature molar heat capacity C = 3R
Solution
At normal temperature
Cv =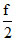 R =
R = 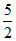 R
R
At any temperature
Cp - Cv =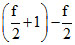 R = R
R = R
From process T = k1V2
and
Ideal gas equations PV = nRT
We have PV-1 = constant ⇒ x = - 1
⇒ C = Cv +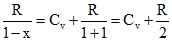
At normal temperature
C =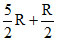 = 3R
= 3R
Cv =
At any temperature
Cp - Cv =
From process T = k1V2
and
Ideal gas equations PV = nRT
We have PV-1 = constant ⇒ x = - 1
⇒ C = Cv +
At normal temperature
C =
Create a free account to view solution
View Solution FreeMore Heat and Thermal Expansion Questions
One mole of an ideal monatomic gas is taken from A to C along the path ABC. The temperature of the gas at A is T0. For t...Certain prefect gas is found to obey PVn = constant during adiabatic process. The volume expansion cofficient at tempera...In a vertical U - tube containing a liquid, the two arms are maintained at different temperatures, t1and t2. The liquid ...A centigrade and a Fahrenheit thermometer are dipped in boiling water. The water temperature is lowered until the Fahren...Two different rods A and B are kept as shown in figure. The variation of temperature of different cross sections with di...