Heat and Thermal ExpansionHard
Question
The reversible expansion of an ideal gas under adiabatic and isothermal conditions is shown in the figure. Which of the following statement(s) is (are) not correct ?
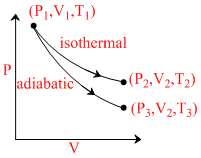

Options
A.T1 = T2
B.T3 > T1
C.Wisothermal > Wadiabatic
D.ᐃUisothermal > ᐃUadiabatic
More Heat and Thermal Expansion Questions
Four containers are filled with monoatomic ideal gases. For each container, the number of moles, the mass of an individu...The temperature of an isotropic cubical solid of length l0 density ρo and coefficient of liner expansion is increas...One of the representatives of Phylum Arthropodais :...The molar specific heat of the the process V ∝ T4 for CH4 at room temperature is : -...Certain prefect gas is found to obey PVn = constant during adiabatic process. The volume expansion cofficient at tempera...