Mole ConceptHard
Question
When 10 mL of ethyl alcohol (density = 0.7893 g mL-1) is mixed with 20 mL of water (density 0.9971 g mL-1) at 25oC, the final solution has a density of 0.9571 g mL-1. The percentage change in total volume on mixing is
Options
A.3.1%
B.2.4%
C.1%
D.None of these
Solution
Total weight of alcohol and water
= 10 × 0.7893 + 20 × 0.9971
Volume of mixture =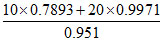 = 29.08 mL
= 29.08 mL
Change in volume = (20 + 10) - 29.08 = 0.92 mL
% change in volume =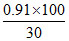 = 3.06% ≈ 3.1%
= 3.06% ≈ 3.1%
= 10 × 0.7893 + 20 × 0.9971
Volume of mixture =
Change in volume = (20 + 10) - 29.08 = 0.92 mL
% change in volume =
Create a free account to view solution
View Solution FreeMore Mole Concept Questions
Which of the following exihibit the weakest intermolecular forces :-...Change in volume of the system does not alter the number of moles in which of the following equilibria ?...How many moles of electron weigh one kilogram :...The empirical formula of a compound is CH2O Its molecular weight is 180. The molecular formula of compound is :...An organic compound contains C, H, and O. If C(%) : H(%) = 6 : 1, what is the simplest formula of the compound, given th...