Mole ConceptHard
Question
How many moles of electron weigh one kilogram :
Options
A.6.023 × 1023
B.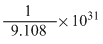
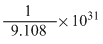
C.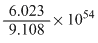
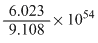
D.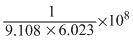
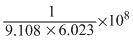
Solution
1 electron = 9.108 × 10-31 kg
∴ 1 Kg =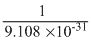 electron
electron
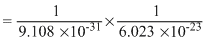 mole electron
mole electron
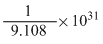
∴ 1 Kg =
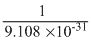 electron
electron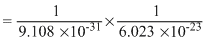 mole electron
mole electron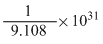
Create a free account to view solution
View Solution FreeMore Mole Concept Questions
Assuming fully decomposed, the volume of CO2 released at STP on heating 9.85g of BaCO3 (Atomic mass of Ba = 137) will be...For preparing 0.1 N solution of a compound from its impure sample sample of which the percentage purity is known, the we...If H2SO4 is formed from it′s elements by taking 6.023 × 1023 atom of ′O′ 5.6 litre of H2 gas and ...According to the collision theory, most molecular collisions do not lead to reaction. Which of the following is/are nece...Which expansion will produce more change in entropy during reversible & isothermal process?...