Mole ConceptHard
Question
The empirical formula of a compound is CH2O Its molecular weight is 180. The molecular formula of compound is :
Options
A.C4HO4
B.C3H6O3
C.C6H12O6
D.C5H10O5
Solution
Empirical formula weight
= 12 + 2 + 16 = 30
n =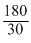 = 6
= 6
Molecular formula = (CH2O)6 = C6H12O6.
= 12 + 2 + 16 = 30
n =
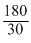 = 6
= 6Molecular formula = (CH2O)6 = C6H12O6.
Create a free account to view solution
View Solution FreeMore Mole Concept Questions
Infection of Ascaris usually occurs by :...The molar concentration of HCl (aq.) is 10-5 M. Which of the following statements are correct.(dsolution = 1 gm /cc)...Mole fraction of the component A in vapour phase is x1 and mole fraction of component A in liquid mixture is x2 (PoA = v...For the reaction 2P + Q → R, 12 mol of P and 8 mol of Q are taken then...The temperature at which the root mean square velocity of SO2 molecules is same as that of O2 molecules at 27oC :-...