Gaseous StateHard
Question
When 2 g of gas A is introduced into an evacuated flask kept at 25oC, the pressure was found to be 1 atomsphere. If 3 g of another gas B is then added to the same flask, the pressure becomes 1.5 atm. Assuming ideal behaviour, the ratio of molecules weights (MA : MB) is
Options
A.1 : 3
B.3 : 1
C.2 : 3
D.3 : 2
Solution
Moles ∝ Pressure
i.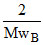 ∝ 1 atm
∝ 1 atm
Pressure of B = 1.5 - 1 = 0.5 atm
ii.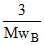 ∝ 0.5 atm
∝ 0.5 atm
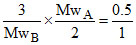
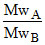 = 0.5 ×
= 0.5 × 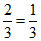
∴ MwA : MwB = 1 : 3
i.
Pressure of B = 1.5 - 1 = 0.5 atm
ii.
∴ MwA : MwB = 1 : 3
Create a free account to view solution
View Solution FreeMore Gaseous State Questions
1 mol of an ideal gas at 27oC is compressed reverssibly from 20 L to 2L. Find out ᐃS :-...A cylinder is filled with a gaseous mixture containing equal masses of CO and N2. The partial pressure ratio is :...The density of neon will be highest at :...Which of the following is not the correct set of pressure and volume at constant temperature and constant moles of gas ?...The curve of pressure volume (PV) against pressure (P) of the gas at a particular temperature is as shown, according to ...