Gaseous StateHard
Question
In van der Waals′ equation of state for a non-ideal gas the term that accounts for intermolecular forces is :
Options
A.(V - b)
B.RT
C.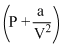
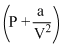
D.(RT)-1
Solution
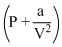 represents the intermolecular forces.
represents the intermolecular forces. Create a free account to view solution
View Solution FreeMore Gaseous State Questions
Select correct statement(s):...When CO2 under high pressure is released from a fire extinguisher, particles of solid CO2 are formed, despite the low su...The temperature of an ideal gas is increased from 120 K to 480 K. If at 120 K the root-mean-square velocity of the gas m...A vessel of volume 5 litre contains 1.4 g of nitrogen at a temperature 1800 K. The pressure of the gas if 30% of its mol...A certain gas takes three times as long to effuse out as helium. Its molecular mass will be:...