Gaseous StateHard
Question
The rate of diffusion of SO2, CO2, PCl3 and SO3 are in the following order
Options
A.PCl3 > SO3 > SO2 > CO2
B.CO2 > SO2 > PCl3 > SO3
C.SO2 > SO3 > PCl3 > CO2
D.CO2 > SO2 > SO3 > PCl3
Solution
Rate ∝ 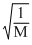 . The smaller the value of M the more is the rate of diffusion
. The smaller the value of M the more is the rate of diffusion
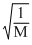 . The smaller the value of M the more is the rate of diffusion
. The smaller the value of M the more is the rate of diffusion Create a free account to view solution
View Solution FreeMore Gaseous State Questions
Consider the following statements: The coefficient B in the virial equation of state(i) is independent of temperature(ii...50 mL of each gas A and of gas B takes 200 and 150 seconds respectively for effusing through a pinhole under the similar...Two flasks of equal volume are connected by a narrow tube (of negligible volume) all at 27oC and contain 0.70 moles of H...A thin balloon filled with air at 47oC has a volume of 3 litre. If on placing it in a cooled room its volume becomes 2.7...The compressibility of a gas is less than unity at STP. Therefore,...