Heat and Thermal ExpansionHard
Question
A gas expands such that its initial and final temperature are equal. Also, The process followed by the gas traces a straight line on the P - V diagram :-
Options
A.The temperature of the gas remains constants throughout.
B.The temperature of the gas first increase and then decrease
C.The temperature of the gas first decrease and then increase.
D.The straight line has a negative slope.
Solution
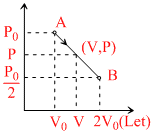
Let V0 = intial volume = 2V0 = final volume
V = volume of any state
then
⇒ nRT = PV = V
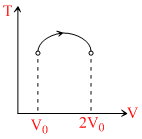
∴ T - V curve is a parabola with vertex above.
Hence temperature first increase then decrease
Create a free account to view solution
View Solution FreeMore Heat and Thermal Expansion Questions
A gas takes part in two processes in which it is heated from the same initial state 1 to the same 1 to the same final te...A refrigerator converts 100 g of water at 25oC into ice at - 10oC in one hour and 50 minutes. The quantity of heat remov...Hailstone at 0oC falls from a height of 1 km on an insulating surface converting whole of its kinetic energy into heat. ...5n, n and 5n moles of a monoatomic, diatomic and non - linear polyatomic gases (which do not react chemically with each ...Two substance A and B of equal mass m are heated by uniform rate of 6 cal s-1 similar conditions. A graph between temper...