Heat and Thermal ExpansionHard
Question
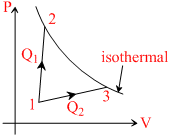
A gas takes part in two processes in which it is heated from the same initial state 1 to the same 1 to the same final temperature. The processes are shown on the P-V diagram by the straight line 1 - 2 and 1 - 3.2 and 3 are the points on the same isothermal curve. Q1 and Q2 are the heat transfer along the two processes. Then :-

Options
A. Q1 = Q2
B. Q1 < Q2
C. Q1 > Q2
D.insufficient data
Solution
Work done in process 1 - 3 is greater than that in process 1 - 2. while change in internal energy is same for both processes ∴ Q1 > Q2
Create a free account to view solution
View Solution FreeMore Heat and Thermal Expansion Questions
Figure shows the variation of the internal energy U with density ρ of one mole of an ideal monatomic gas for thermo...Radius of a conductor increase informally from left end to right end as shown in Fig. Material of the conductor is isotr...Certain prefect gas is found to obey PVn = constant during adiabatic process. The volume expansion cofficient at tempera...Two long, thin solid cylinders are evidential in size, but they are of different substances with two different thermal c...An ice box made of styrofoam (thermal conductivity = 0.01 Jm-1 s-1 K-1) is used to keep liquids cool. It has a total wal...