Heat and Thermal ExpansionHard
Question
5n, n and 5n moles of a monoatomic, diatomic and non - linear polyatomic gases (which do not react chemically with each other ) are mixed at room temperature. The equivalent degree of freedom for the mixture is : -
Options
A.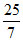
B.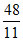
C.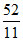
D.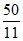
More Heat and Thermal Expansion Questions
When water is heated from 0oC to 4oC and CP and CV are specific heats at constant pressure and constant volume respectiv...A point source of heat of power P is placed at the center of a spherical shell of mean radius R the material of the shel...A block body emits at the rate P when its temperature is T. At this temperature the wavelength at which the radiation ha...Figure shows the variation of the internal energy U with density ρ of one mole of an ideal monatomic gas for thermo...n moles of an ideal triatomic linear gas undergoes a process in which the temperature changes with volume as T = k1V2 wh...