Heat and Thermal ExpansionHard
Question
A refrigerator converts 100 g of water at 25oC into ice at - 10oC in one hour and 50 minutes. The quantity of heat removed per minute is : - (Specific heat of ice = 0.5 cal/goC, latent heat of fusion = 80 cal/g )
Options
A.50 cal
B.100 cal
C.200 cal
D.75 cal
Solution
Heat removed in cooling water from 25oC to 0oC = 100 × 1 × 25 = 2500 cal
Heat removed in converting water into ice at 0oC = 10 × 80 = 8000 cal
Heat removed in cooling ice from
0o to - 15oC = 100 × 0.5 × 10 = 500 cal
Total heat removed in 1 hr 50 min = 2500 + 8000 + 500 = 11000 cal
Heat removed per minute =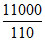 = 100 cal / min
= 100 cal / min
Heat removed in converting water into ice at 0oC = 10 × 80 = 8000 cal
Heat removed in cooling ice from
0o to - 15oC = 100 × 0.5 × 10 = 500 cal
Total heat removed in 1 hr 50 min = 2500 + 8000 + 500 = 11000 cal
Heat removed per minute =
Create a free account to view solution
View Solution FreeMore Heat and Thermal Expansion Questions
A metallic sphere having radius 0.08 m and mass m = 10 kg is heated to a temperature of 227oC and suspended inside a box...During experiment, an ideal gas is found to obey a condition P2/ρ = constant [ρ density of the gas]. The gas i...If the intermolecular force vanish away, the volume occupied by the molecules contained in 4.5 kg water at standard temp...Three rods of the same dimensions have thermal conductivities 3k, 2k and k. They are arranged as shown, with their ends ...A steel scale is to be prepared such that the millimeter intervals are to be accurate4 within 6 × 10-5 mm.The maxim...