Chemical EquilibriumHard
Question
For the equilibrium at 298 K; N2O4(g) ⇋ 2NO2(g); G-N2O4 = 100 kJ mol-1 and G-NO2 = 50 kJ mol-1. If 5 mol of N2O4 and 2 moles of NO2 are taken initially in one liter container than which statement are correct.
Options
A.Reaction proceeds in forward direction
B.Kc = 1
C.ᐃG = - 0.55 KJ, ᐃG- = 0
D.At equilibrium [N2O4] = 4.84 M and [NO2] = 0.212 M
Solution
ᐃG = ᐃG- + 2.303 RT log Q
ᐃG = 2 × G-NO2 - G-N2O4 = 2 × 50 - 100 = 0
∴ ᐃG = 0 + 2.303 × 8.314 × 10-3 × 298 log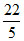
= 0 - 0.55 kJ
∴ ᐃG = -0.55 kJ, i.e., reaction proceed in forward direction
Also ᐃG- = 0 = 2.303 RT log K ∴ K = 1
Now, N2O4 - 2NO2
5 2
5 - x 2 + 2x
∴ Kp =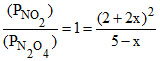 or x = 0.106
or x = 0.106
ᐃG = 2 × G-NO2 - G-N2O4 = 2 × 50 - 100 = 0
∴ ᐃG = 0 + 2.303 × 8.314 × 10-3 × 298 log
= 0 - 0.55 kJ
∴ ᐃG = -0.55 kJ, i.e., reaction proceed in forward direction
Also ᐃG- = 0 = 2.303 RT log K ∴ K = 1
Now, N2O4 - 2NO2
5 2
5 - x 2 + 2x
∴ Kp =
Create a free account to view solution
View Solution FreeMore Chemical Equilibrium Questions
One mole each of A and B and 3 moles each of C and D are placed in 1 L flask. If equilibrium constant is 2.25 for the re...For the reaction A + B$\rightleftharpoons$C + D, the initial concentration of A and B is equal, but the equilibrium conc...The equilibrium constant Kp1 and Kp2 for the reactions X ⇋ 2Y and Z ⇋ P + Q ; respectively are in the ratio ...A 20 litre container at 400 K contains CO2(g) at pressure 0.4 atm and an excess of SrO (neglect the volume of solid SrO)...The equilibrium constants for the reaction A2 $\rightleftharpoons$2A at 500 K and 1000 K are 1 × 10–10 and 1 × 10–5, res...