Chemical EquilibriumHard
Question
The equilibrium constant Kp1 and Kp2 for the reactions X ⇋ 2Y and Z ⇋ P + Q ; respectively are in the ratio of 1 : 9. If the degree of dissociation of X and Z be equal then calculate value of 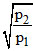 . Where p1 and p2 are total pressure of given two reactions at their equilibrium.
. Where p1 and p2 are total pressure of given two reactions at their equilibrium.
Options
A.9
B.1/9
C.6
D.1/6
Solution
1/6X ⇋ 2Y
t0 1 0
teq 1-α 2α
Kp1 =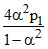
Z ⇋ P + Q
1 0 0
1-α α α
Kp2 =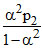
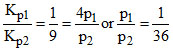
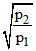 = 6
= 6
t0 1 0
teq 1-α 2α
Kp1 =
Z ⇋ P + Q
1 0 0
1-α α α
Kp2 =
Create a free account to view solution
View Solution FreeMore Chemical Equilibrium Questions
At 0°C and 1 atm pressure, 1 L of N2O4 decomposes to NO2 according to the equation N2O4(g) $\rightleftharpoons$2NO2 (g)....Densities of diamond and graphite are 3.5 and 2.3 g/mL. C (diamond) ⇋ C (graphite) ᐃrH = - 1.9 kJ/molefavour...For a gaseous equilibrium 2A(g) $\rightleftharpoons$2B(g) + C(g), KP has a value 1.8 at 700 K. The value of KC for the e...X2(g) + Y2(g) ⇋ 2XY(g) reaction was studied at a certain temperature. In the beginning 1 mole of X2 was taken in a...A reaction at 300 K with ΔGo = −1743 J consists of 3 moles of A(g), 6 moles of B(g) and 3 moles of C(g). If A, B and C a...