Chemical BondingHard
Question
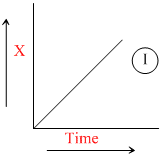
The graph between concentration (X) of the product and time of the reaction A → B is of the type I. Hence, graph between - 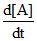 and time will be of the type:
and time will be of the type:

Options
A.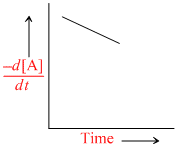

B.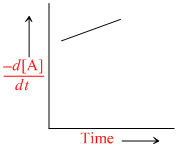

C.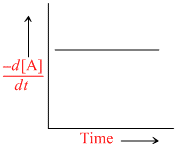

D.

Solution
Since [X] with time is increasing uniformly so change in concentration of A or B, i.e, 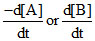 is constant.
is constant.
Create a free account to view solution
View Solution FreeMore Chemical Bonding Questions
The bond angle between H-O-H in ice is closest to...The type of hybrid orbitals used by the chlorine atom in ClO2- is :...Give the correct reaction :- (a) NaNO2 NO(g)(b) HNO3 N2O5(c) P4 + NaOH → PH3 + Na3PO4(d) (NH4)2Cr2O7 N2O + Cr2O3 +...In which pair of species the hybrid state of B is sp3 :-...A magnetic moment of 1.73 BM will be shown by one among the following :-...