Chemical BondingHard
Question
The type of hybrid orbitals used by the chlorine atom in ClO2- is :
Options
A.sp3
B.sp2
C.sp
D.None of these
Solution
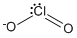 Sum of σ bond pair + lone pair = 4 (sp3 - hybridised)
Sum of σ bond pair + lone pair = 4 (sp3 - hybridised) Create a free account to view solution
View Solution FreeMore Chemical Bonding Questions
Identify the last stable ion amongst the following :...The states of hybridization of boron and oxygen atoms in boric acid (H3BO3) are respectively...Deduce from the following E - values of half cells, what combination of two half cell would results in a cell with the l...In the reaction NH3 + H2O ⇋ NH2- + H3O+ the conjugate base of NH3 is :-...The O-O bond length in O2, O2[AsF4] and K[O2] is :-...