Chemical BondingHard
Question
A magnetic moment of 1.73 BM will be shown by one among the following :-
Options
A.[CoCl6]4-
B.[Cu(NH3)4]2+
C.[Ni(CN)4]2-
D.TiCl4
Solution
Magnetic moment 1.73 BM
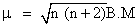
n = no. of unpaired e-
μ = 1.73 =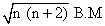
n = 1
• [CoCl6]4- ⇒ Co+2 ; d7
Cl- (weak field ligand) t2g5eg2 unpaired e-= 3
• [Cu(NH3)4]2+ ⇒ Cu+2 ; d9
NH3 Strong field ligand, hybridisation dsp2
• one e- of 3d jumps into 4p subshell.
unpaired e- = 1
• [Ni(CN)4]2- ⇒ Ni+2 ; d8 unpaired e- = 0
CN- = Strong field ligand dsp2
• TiCl4 ⇒ Ti+4 ; do unpaired e- = zero.
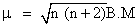
n = no. of unpaired e-
μ = 1.73 =
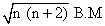
n = 1
• [CoCl6]4- ⇒ Co+2 ; d7
Cl- (weak field ligand) t2g5eg2 unpaired e-= 3
• [Cu(NH3)4]2+ ⇒ Cu+2 ; d9
NH3 Strong field ligand, hybridisation dsp2
• one e- of 3d jumps into 4p subshell.
unpaired e- = 1
• [Ni(CN)4]2- ⇒ Ni+2 ; d8 unpaired e- = 0
CN- = Strong field ligand dsp2
• TiCl4 ⇒ Ti+4 ; do unpaired e- = zero.
Create a free account to view solution
View Solution FreeMore Chemical Bonding Questions
At 27oC, 1 mol gas is allowed to expand reversibly and adiabatically then the final temperature of the system becomes 10...Which one of the following compounds has sp2 - hybridisation ?...The geomety of H2S and its dipole moment are :...CH3 - C ≡ N , Z is :-...Under hydrolytic conditions, the compounds used for preparation of linear polymer and for chain termination, respectivel...