ElectrochemistryHard
Question
The emf of Daniel cell at 298 K is E1Zn|ZnSO4(0.01M)||CuSO4(1.0M)|Cu When the concentration of ZnSo4 is 1.0 M and that of CuSO4 is 0.01 M the emf changed to E2
What in the relation between E1 and E2 ?
What in the relation between E1 and E2 ?
Options
A.E1 = E2
B.E2 = 0 ≠ E2
C.E1 > E2
D.E1< E2
Solution
Using the relatio
Ecell = Eocell -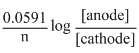
= Eocell -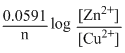
Substituting the given values in two cases.
E1 = E0 -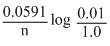
= E0 -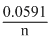 log 10-2
log 10-2
= E0 +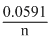 × or (E0 + 0.0591)V
× or (E0 + 0.0591)V
E2 = E0 -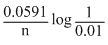
= E0 -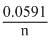 log 102
log 102
= E0 -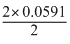 or (E0 - 0.0591)V
or (E0 - 0.0591)V
Thus, E1 > E2
Ecell = Eocell -
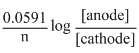
= Eocell -
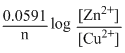
Substituting the given values in two cases.
E1 = E0 -
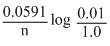
= E0 -
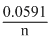 log 10-2
log 10-2= E0 +
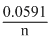 × or (E0 + 0.0591)V
× or (E0 + 0.0591)VE2 = E0 -
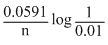
= E0 -
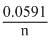 log 102
log 102 = E0 -
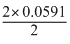 or (E0 - 0.0591)V
or (E0 - 0.0591)VThus, E1 > E2
Create a free account to view solution
View Solution FreeMore Electrochemistry Questions
If the pressure of hydrogen gas is increased from 1 atm. to 100 atm, keeping the hydrogen ion concentration constant at ...Expermentally it was found that a metal oxide has formula M0.98O. Metal M, is present as M2+and M3+ in its oxide. Fracti...Which of the following statements is true for the electrochemical, Daniel cell ?...Red hot carbon will remove oxygen from the oxide AO and BO but not from MO, while B will remove oxygen from AO. The acti...A line L : y = mx + 3 meets y - axis at E(0,3) and the arc of the parabola y2 = 16x, 0 ≤ y ≤ 6 at the point ...