ElectrochemistryHard
Question
A 0.5 M NaOH solution offers a resistance of 31.6 ohm in a conducivity cell at roomtemperature. What shall be the apporiximate molar condcutance of this NaOH solution if cell constant of the cell is 0.367 cm-1
Options
A.234 S cm2mole-1
B.23.2 S cm2mole-1
C.4645 S cm2mole-1
D.5464 S cm2mole-1
Solution
Here, R = 31.6ohm
∴ C =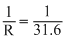 ohm-1 = 0.0316 ohm-1
ohm-1 = 0.0316 ohm-1
Specific conductance
= conductance × cell constant
= 0.0316ohm-1 × 0.367cm-1
= 0.0116ohm-1cm-1
Now ,molar concentration = 0.5 m (given)
= 0.5 × 10-3 mole cm-3
∴ Molar conductance =
=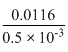 = 23.2S cm2mol-1
= 23.2S cm2mol-1
∴ C =
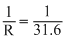 ohm-1 = 0.0316 ohm-1
ohm-1 = 0.0316 ohm-1Specific conductance
= conductance × cell constant
= 0.0316ohm-1 × 0.367cm-1
= 0.0116ohm-1cm-1
Now ,molar concentration = 0.5 m (given)
= 0.5 × 10-3 mole cm-3
∴ Molar conductance =

=
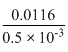 = 23.2S cm2mol-1
= 23.2S cm2mol-1Create a free account to view solution
View Solution FreeMore Electrochemistry Questions
Zinc granules are added in excess to a 500 ml of 1.0 M nickel nitrate solution at 25°C until the equilibrium is reached....Given the ionic conductance of , K+, and Na+ are 74, 50 and 73 cm2 ohm-1 eq-1, respectively. The equivalent conductance ...Equivalent conductance of an electrolyte containing NaF at infinite dilution is 90.1 Ohm-1 cm2 If NaF is replaced by KF ...When a lead storage battery is discharged :...An ion is a reduced to the element when it absorbs 6 × 1020 electrons. The number of equivalents of the ion is...