ElectrochemistryHard
Question
Given the ionic equivalent conductivities for the following ions:
λoeq K+ = 73.5 cm2 ohm-1 eq-1
λoeq Al3+ = 149 cm2 ohm-1 eq-1
λoeq SO42 = 85.8 cm2 ohm-1 eq-1
The Λoeq for potash alum (K2SO4.Al2(SO4)3.24H2O) is:
λoeq K+ = 73.5 cm2 ohm-1 eq-1
λoeq Al3+ = 149 cm2 ohm-1 eq-1
λoeq SO42 = 85.8 cm2 ohm-1 eq-1
The Λoeq for potash alum (K2SO4.Al2(SO4)3.24H2O) is:
Options
A.- 245.92
B.348.3
C.368.2
D.108.52
Solution
(Eq = Charge on the ion/Total charge)
[A+] =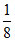 mol × 2 =
mol × 2 = 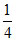 mol =
mol = 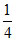 Eq
Eq
[Al3+] =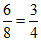 Eq
Eq
[SO42-] =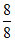 = 1Eq
= 1Eq
Λoeq K2SO4.Al2(SO4)3. 24H2O
= λoeq(K+) + λoeq (Al+3) + λoeq(SO42-)
=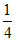 × 73.5 + 189 ×
× 73.5 + 189 × 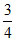 + 85.8 × 1
+ 85.8 × 1
= 18.375 + 141.75 + 85.8
= 249.92
[A+] =
[Al3+] =
[SO42-] =
Λoeq K2SO4.Al2(SO4)3. 24H2O
= λoeq(K+) + λoeq (Al+3) + λoeq(SO42-)
=
= 18.375 + 141.75 + 85.8
= 249.92
Create a free account to view solution
View Solution FreeMore Electrochemistry Questions
Beryllium is placed above magnesium in the group II. Beryllium dust, therefore, when added to MgCl2 solution will...During the electrolysis of 0.1 M CuSO4 solution using copper electrodes, a depletion of [Cu++] occurs near the cathode w...Assertion :- BeSO4 is more soluble in water than BaSO4.Reason :- In BeSO4 H.E. exceeds its L.E....What is the pH of 0.01 m glycine solution ? for glycine, Ka1 = 4.5 × 10-3 and Ka2 = 1.7 × 10-10 at 298K ?...Resistance of 0.2 M solution of an electrolyte is 50 Ω. The specific conductance of the solution is 1.4 S m−1. The resis...