Mole ConceptHard
Question
A nvesel conains 14 g (moles) of hydrogen and 96 g (3 moles) of oxygen at ATP. Chemical reaction induced by passing electric spark in the vessel till one of the gasses is consumed. The temperature is brought back to it’s starting value k. The pressure in the vessel is -
Options
A.0.1 atm
B.0.2 atm
C.0.3 atm
D.0.4 atm
Solution
When electric spark is passed, hydrogen reacts with oxygen to from water (H2O). Each gram of hydrogen reacts with eight grams of oxygen. Thus 96 g of oxygen will be totally consumed with 12 g hydrogen.
The gas left in the vessel will be 2g of hydrogen i.e. number of moles μ =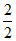 = 1.
= 1.
Using PV = μRT ⇒ P ∝ μ ⇒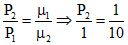 ⇒ P2 = 0.1 atm
⇒ P2 = 0.1 atm
The gas left in the vessel will be 2g of hydrogen i.e. number of moles μ =
Using PV = μRT ⇒ P ∝ μ ⇒
Create a free account to view solution
View Solution FreeMore Mole Concept Questions
The density of air is 0.001293 g/cm3 at STP. Identify which of the following statement is correct...100 gm impure CaCO3 on heating gives 5.6 lt. CO2 gas at STP. Find the percentage of calcium in the lime stone sample. [A...A 5L vessel contains 2.8 g of N2. When heated to 1800 K, 30% molecules are dissociated into atoms....If we consider that , in place of ; mass of carbon atom is taken to be the relative atomic mass unit, the mass of one mo...The vapour density of a sample of SO3 gas is 28. Its degree of dissociation into SO2 and O2 is...