Mole ConceptHard
Question
The density of air is 0.001293 g/cm3 at STP. Identify which of the following statement is correct
Options
A.Vapour density is 14.48
B.Molecular weight is 28.96
C.Vapour density is 0.001293 g/cm3
D.Vapour density and molecular weight cannot be determined.
Solution
(A) and (B) Explanation : M. Wt. = 0.001293 × 22400 = 28.96
M.Wt. = d × volume of 1 mole of gas at STP
V. D =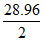 = 14.48
= 14.48
So (A) and (B) are correct answer.
M.Wt. = d × volume of 1 mole of gas at STP
V. D =
So (A) and (B) are correct answer.
Create a free account to view solution
View Solution FreeMore Mole Concept Questions
Which of the following has the smallest number of molecules :-...If NA is Avogadro′s number then number of valence electrons in 4.2 g of nitride ions (N3-)...Some water is added to a mixture of 250 ml of 0.6 M HCl and 750 ml of 0.2 M HCl to obtain 0.25 M solution of HCl as a re...Which of the following is dependent on temperature?...For the complete combustion of 0.2 moles of the sugar, how many moles of oxygen is required ?...