Mole ConceptHard
Question
1.2575 g sample of [Cr(NH3)6]SO4Cl(Mw = 251.5) is dissolved to prepare 250mL solution showing and osmotic pressure of 1.478 atm of Hg at 27oC. Which of the following statements is/are correct about this solution?
Options
A.Each molecule furnishes three ions in solution.
B.The Van′t Hoff factor is = 3.
C.The equilibrium molarity of [Cr(NH3)6]SO4Cl = 0.
D.The equilibrium molarity of [Cr(NH3)6]3+ = 0.02 M.
Solution
Molarity 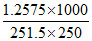 = 0.02 M, π = CRT
= 0.02 M, π = CRT
∴ πobs = 0.02 × 0.0821 × 300 = 0.4926 atm
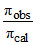 = i =
= i = 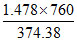 = 3
= 3
∵ α =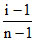 and = 1
and = 1
∴ n = 3
Also,
[Cr(NH3)6]SO4.Cl ⇋ [Cr(NH3)63+] + SO42- + Cl-
0.02 0 0 0
Eq conc 0 0.02 0.02 0.02
∴ πobs = 0.02 × 0.0821 × 300 = 0.4926 atm
∵ α =
∴ n = 3
Also,
[Cr(NH3)6]SO4.Cl ⇋ [Cr(NH3)63+] + SO42- + Cl-
0.02 0 0 0
Eq conc 0 0.02 0.02 0.02
Create a free account to view solution
View Solution FreeMore Mole Concept Questions
In the reaction. 2Al(s) + 6HCl(s) → 2Al3+(aq) + 6Cl-(aq) + 3H2(g),...1 volume of gaseous compound consisting C, H, O on complete combustion in presence of 2.5 volume of O2 gives 2 vol. of s...Iron forms two oxides. If for the same mass of iron, the mass of oxygen combined in the first oxide is two-third of the ...A gaseous mixture contains 70% N2 and 30% unknown gas by volume. If the average molecular mass of gaseous mixture is 37....The Ksp of Ag2CrO4, AgCl, AgBr and AgI are respectively, 1.1 × 10-12, 1.8 × 10-10,. 5.0 × 10-13, 8.3 ...