Mole ConceptHard
Question
1 volume of gaseous compound consisting C, H, O on complete combustion in presence of 2.5 volume of O2 gives 2 vol. of steam and 2 vol. of CO2, what is the formula of the compound if all measurements are made at NTP ?
Options
A.C2H4O
B.CH3O
C.C2H2O
D.C2H3O2
Solution
CxHyOz + 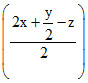 O2 → xCO2(g) +
O2 → xCO2(g) + 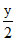 H2O(g)
H2O(g)
x +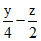 = 2.5
= 2.5
x = 2,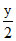 = 2 ⇒ y = 4
= 2 ⇒ y = 4
2 + 1 -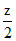 = 2.5 ⇒ z = 1
= 2.5 ⇒ z = 1
C2H4O
x +
x = 2,
2 + 1 -
C2H4O
Create a free account to view solution
View Solution FreeMore Mole Concept Questions
In which of the following ways does an activated complex differ from an ordinary molecule ?...In a compound C, H and N atoms are present in 9 : 1 : 35 by weight. Molecular weight of compound is 108. Molecular formu...A mixture of ethylene and excess of H2 had a pressure of 600 mm Hg. The mixture was passed over nickel catalyst to conve...Given the polymers (i) Nylon 6,6 (ii) Buna-S, (iii) Polythene, arrange these in increasing order of their inter-molecula...Calculate the mass percent (w/w) of sulphuric acid in a solution prepared by dissolving 4 g of sulphur trioxide in a 100...