Mole ConceptHard
Question
In Duma′s method of estimation of nitrogen 0.35 g of an organic compound gave 55 mL of nitrogen collected at 300 K temperature and 715 mm pressure. The percentage composition of nitrogen in the compound will be (Aqueous tension at 300 K = 15 mm)
Options
A.16.45
B.17.45
C.14.45
D.15.45
Solution
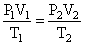
Where, P2 = pressure of N2 at STP = 760 mm
T2 = Temperature of N2 at STP = 273 K
V2 = ?
Volume of N 2 at STP (By gas equation)
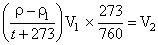
Where, P1 = ρ - ρ1
ρ = 715 mm (pressure at which N2 collected).
ρ1 = aqueous tension of water = 15 mm
T1 = t + 273 = 300 K
V1 = 55 mL = volume of moist nitrogen in nitrometer
∴
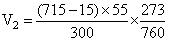 = 46.098 mL
= 46.098 mL% of nitrogen in given compound =
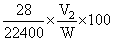
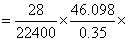 100 = 16.45%
100 = 16.45% Create a free account to view solution
View Solution FreeMore Mole Concept Questions
A volume of 10 ml of gaseous C4Hx exactly requires 55 ml O2 for complete combustion. The value of ‘x’ is...If we consider that , in place of ; mass of carbon atom is taken to be the relative atomic mass unit, the mass of one mo...A mixture of ethylene and excess of H2 had a pressure of 600 mm Hg. The mixture was passed over nickel catalyst to conve...A 1.50 g sample of type metal (an alloy of Sn, Pb, Cu and Sb) is dissolved in nitric acid, and met stannic acid, H2SnO3,...When 0.03 L of a mixture of hydrogen and oxygen was exploded, 0.003 L of oxygen remained. The initial mixture contains (...