Ionic EquilibriumHard
Question
A buffer solution is prepared in which the concentration of NH3 is 0.30 M and the concentration of NH4+ is 0.20 M. If the equilibrium constant, Kb for NH 3 equals 1.8 × 10-10 , what is the pH of this solution ? (log 2.7 = 0.43)
Options
A.9.43
B.11.72
C.8.73
D.9.08
Solution
pOH = pKb + log 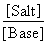
= - log Kb + log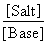
= - log 1.8 × 10-5 + log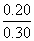
= 5 - 0.25 + (- 0.176)
= 4.75 - 0.176 = 4.57
∴ pH = 14 - 4.57 = 9.43
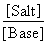
= - log Kb + log
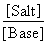
= - log 1.8 × 10-5 + log
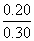
= 5 - 0.25 + (- 0.176)
= 4.75 - 0.176 = 4.57
∴ pH = 14 - 4.57 = 9.43
Create a free account to view solution
View Solution FreeMore Ionic Equilibrium Questions
The pKa of a weak acid, HA, is 4.80. The pKb of a weak base, BOH, is 4.78. The pH of an aqueous solution of the correspo...Ag+ + NH3 $\rightleftharpoons$ [Ag(NH3)+]; K1 = 1.6 × 103[Ag(NH3)+] + NH3 $\rightleftharpoons$ [Ag(NH3)2+]; K2 = 6.8 ×...If 50 ml of 0.2 M KOH is added to 40 ml of 0.5 M HCOOH. the pH of the resulting solution is: (Ka = 1.8 × 10-4, log ...Which of the following are true for an acid-base titration ?...From separate solutions of for sodium salts NaW, NaX, NaY and NaZ has pH 7.0, 9.0 10.0 and 11.0 respectively. When each ...