Ionic EquilibriumHard
Question
If 50 ml of 0.2 M KOH is added to 40 ml of 0.5 M HCOOH. the pH of the resulting solution is:
(Ka = 1.8 × 10-4, log 18 = 1.26)
(Ka = 1.8 × 10-4, log 18 = 1.26)
Options
A.3.74
B.5.64
C.7.57
D.3.42
Solution
HCOOH + KOH → HCOOK + H2O
milimole 20 10
10 - 10
pH = pKa + log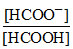 = 3.74 + log
= 3.74 + log 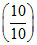 ⇒ pH = 3.74
⇒ pH = 3.74
milimole 20 10
10 - 10
pH = pKa + log
Create a free account to view solution
View Solution FreeMore Ionic Equilibrium Questions
At 30oC the solubility of Ag2CO3 (KSP = 8 × 10-12) would be greatest in one litre of:...Acetic acid and propionic acid have Ka values 1.75 × 10-5 and 1.3 × 10-5 respectively at a certain temperature...The best indicator for the detection of end point in titration of a weak acid and a strong base is :...pH value of which of the following is not equal to one :...In which of the following reactions, the concentration of the product is hither than theconcentration of the product is ...