Chemical BondingHard
Question
The correct order of increasing bond length of C - H, C - O, C - C and C = C is
Options
A.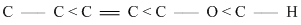
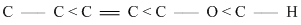
B.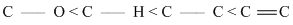
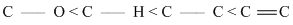
C.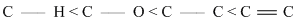
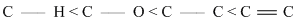
D.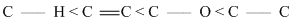
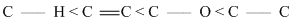
Solution
C - H : 0.109 nm
C = C : 0.134 nm
C - O : 0.143 nm
C - C : 0.154 nm
∴ Bond length order is
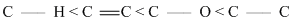
C = C : 0.134 nm
C - O : 0.143 nm
C - C : 0.154 nm
∴ Bond length order is
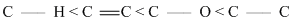
Create a free account to view solution
View Solution FreeMore Chemical Bonding Questions
Among $H_{2}\text{ }S,H_{2}O,{NF}_{3},{NH}_{3}$ and ${CHCl}_{3}$, identify the molecule $(X)$ with lowest dipole moment ...Thermite reduction is not used for commercial extraction of respective metal from which of the following oxides -...In which of the following pairs the two species are not isostructural ?...Pair of species among the following having same bond order as well as paramagnetic character will be-...The dipole moment of AX3, BX3 and CY3 are 4.97 × 10-30, 0.60 × 10-30 and 0.00 C.M. respectively, then the shap...