Chemical BondingHard
Question
In which of the following pairs the two species are not isostructural ?
Options
A.AlF63- and SF6
B.CO32- and NO3-
C.PCl4+ and SiCl4
D.PF5 and BrF5
Solution
(1) AlF63- and SF6 → both are octahedral.
(2) CO32- and NO3 → both are triangular planar
(3) PCl4+ and SiCl4 → both are tetrahedral
(4) PF5 and BrF5 → PF5 has 5bp, 0 lp (sp3d)Structure is trigonal bipyramidal
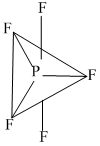
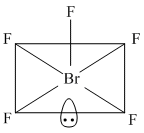
BrF5 has 5bp & 1 lp (sp3d2) and structure issquare pyramidal
(2) CO32- and NO3 → both are triangular planar
(3) PCl4+ and SiCl4 → both are tetrahedral
(4) PF5 and BrF5 → PF5 has 5bp, 0 lp (sp3d)Structure is trigonal bipyramidal

BrF5 has 5bp & 1 lp (sp3d2) and structure issquare pyramidal

Create a free account to view solution
View Solution FreeMore Chemical Bonding Questions
Which of the following molecules has the maximum dipole moment?...The structure of IF7 is :...Which of the following does not contain any coordinate bond ?...Some quantity of water is contained in a container as shown in figure. As neon is added to this system at constant press...Match the following Ore Chemical Formula(A) Limonite (i) Fe2O3.3H2O(B) Cryolite (ii) Cu(OH)2.2CuCO3(C) Calamine (iii) Na...