Chemical BondingHard
Question
The dipole moment of AX3, BX3 and CY3 are 4.97 × 10-30, 0.60 × 10-30 and 0.00 C.M. respectively, then the shape of molecules may be
Options
A.Pyramidal, T-shape, Trigonal planar
B.Pyramidal, pyramidal, T-shape
C.Pyramidal, pyramidal, Trigonal planar
D.Pyramidal, pyramidal, linear
Solution
Since the dipole moment of CY3 = 0. It means C in CY3 is sp3 hybridisation
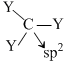 μnet = 0
μnet = 0
Trigonal planar
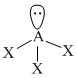 μnet ≠ 0
μnet ≠ 0
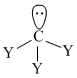
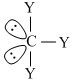
Trigonal pyramidal
Trigonal pyramidal T-shape μnet ≠ 0
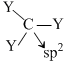 μnet = 0
μnet = 0Trigonal planar
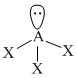 μnet ≠ 0
μnet ≠ 0Trigonal pyramidal


Trigonal pyramidal T-shape μnet ≠ 0
Create a free account to view solution
View Solution FreeMore Chemical Bonding Questions
Which one of the following molecules contains no π bond ?...Given below are some of the statements about Mn and ${Mn}_{2}O_{7}$. Identify the correct statementsA). Mn forms the oxi...The ion that is isorlrctronic with CO is :...The correct statement(s) regarding, (i) HClO, (ii) HClO2, (iii) HClO3 and (iv) HClO4, is(are)...For the hypothetical reactionA2(g) + B2(g) ⇋ 2AB(g)ᐃrGo and ᐃrSo are 20 kJ/mol and -20Jk-1 mol-1 respe...