ElectrochemistryHard
Question
Expermentally it was found that a metal oxide has formula M0.98O. Metal M, is present as M2+and M3+ in its oxide. Fraction of the metal which exists as M3+ would be :-
Options
A.7.01%
B.4.08%
C.6.05%
D.5.08%
Solution
Let no. of oxygen atom = 100
∴ no. of metal atom = 98
if no. of M+2 ion = x
then no. of M+3 ion = (98 - x)
Since compound is electrically neutral
x × (12) + (98 - x)x + 3 + 100 × (-2) = 0
2x + 98 × 3 - 3x - 200 = 0
294 - 200 - x = 0
x = 94
∴ No. of M+2 ion = 94
No. of M+3 ion = 98 - 94 = 4
∴ Fraction of metal which exists as M+3would be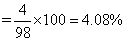
∴ no. of metal atom = 98
if no. of M+2 ion = x
then no. of M+3 ion = (98 - x)
Since compound is electrically neutral
x × (12) + (98 - x)x + 3 + 100 × (-2) = 0
2x + 98 × 3 - 3x - 200 = 0
294 - 200 - x = 0
x = 94
∴ No. of M+2 ion = 94
No. of M+3 ion = 98 - 94 = 4
∴ Fraction of metal which exists as M+3would be
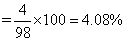
Create a free account to view solution
View Solution FreeMore Electrochemistry Questions
Which of the following facts is not true ?...The electric charge for electrode deposition of one gram equivalent of a substance is :...The correct order of equivalent conductance at infinte dilution of LiCl, NaCl is :...In the following chemical reaction :Ag2O + H2O + 2e- → 2Ag + 2OH-...Mark out the correct statement(s) regarding electrolytic molar conductivity....